
- Age: 8+
- Time: 30
- Setup: 5 min, Activity: 20 min, Cleanup: 5 min
- Materials: $15
Put your construction skills to the test as you race against time and try tricky challenges to build candy crystals!
Download PDF-
what you need
WHAT YOU NEED
Materials:
- 2 different colors of candy, 13 pieces each (wrapped candy like Starburst is best)
- Printouts
- Paper grid
- Data Sheet
- Scoring Guide (for reference)
Equipment
- Stopwatch
- Camera (smartphone preferred)
Note: This activity can be done solo, but it’s fun to do with others. Discussion and competition is encouraged!
-
What To Do
WHAT TO DO
This activity explores different variations of a process scientists use to build a crystal material. To model this, candy will be used to represent atoms. Atoms are the tiny building blocks that make up everything around us.
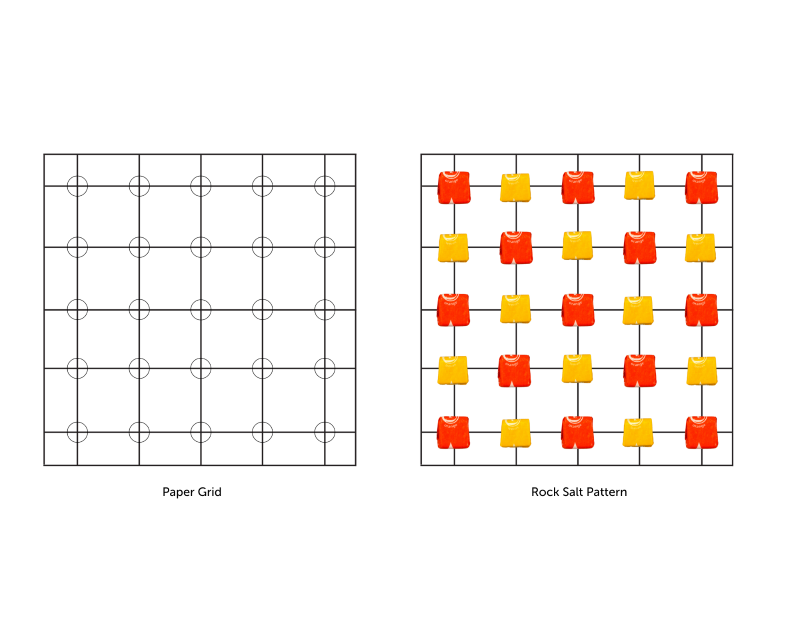
The example crystal structure you will be modeling is commonly called “rock salt,” and it is made from two different kinds of atoms. Below is a picture of what a small rock salt crystal looks like up close. Now imagine zooming in and slicing it into a layer that is one atom thick. If you did, you would see that the atoms in that layer form a crystalline lattice pattern shown in the diagram on the right. This is the structure that you will build.

Stage 1: Initial Crystal Build
- Set the paper grid in front of you to guide your construction. Place the two colors of candy in separate piles off to the side. These are the "atoms" you will use to build your rock salt.
- Get your stopwatch ready and GO! Time how long it takes you to carefully construct the rock salt model.
- Be sure each candy atom covers the small circle at each intersection point.
- Refer to the diagram below to ensure your crystal structure matches the correct pattern. Your goal is to try to build this exact same structure for each method in Stage 2.
- On the Data Sheet, record the time it took to build your rock salt crystal. Take a picture of your final result for reference in the next challenges. Clear the candy atoms from the lattice and place them back into their original piles.
Stage 2: Experimenting with Different Build Conditions
Now try to build the same rock salt crystal structure using a variety of different conditions - just like materials scientists do! As you experiment, some of your atoms might not land in the right place. At the end of each round, count these misplaced atoms. In real crystals, these are called "defects."
For each and every build, be sure to:
- Use a stopwatch to measure your build time and write it on the Data Sheet.
- Keep each atom where it lands. Once your fingers let go of a candy piece, do not move it. You'll learn why this is important...And also part of the fun!
- Take a picture of each finished structure so it can be used for reference later.
- Count the number of defects. (Scroll down to see the Scoring Guide at the bottom of the page.)
- Put your candy atoms back in their original piles before starting the next round.
- If you have two people and enough supplies, build at the same time and have a friendly competition!
- Getting Energized: Build the structure as fast as possible.
- Add speed to your building process! Scientists sometimes use methods to build materials very quickly, too.
- What was your time? How many defects did you have? Record your results on the Data Sheet.
- Slowing Things Down: Place one hand behind your back and recreate the same structure using a one-handed method.
- Scientists sometimes choose to build materials using methods that give them more control. Why do you think that is?
- Was your strategy the same as when using two hands? How do your build time and number of defects compare?
- Dropping In: Drop each piece of candy from about 6 inches above the grid. Observe how the candies land and try again to make the same rock salt structure.
- Scientists can't always control exactly where each atom goes, but they do their best with the tools and methods they have.
- How well can you control the landing location of each candy atom? What do you notice about the spacing between atoms? Did you use any strategies that helped?
- Going in Blind: Close your eyes and build the structure.
- Because atoms are so small, scientists can't see how and where they travel when a material is being created. This build method mimics the non-visible aspect of making real materials.
- Plan how to build your material without using sight, and try it out. Don't peek!
- Total Chaos: Gather all the candy into your hands. Dump it on the mat at once.
- No need for a stopwatch here (the time is ~0 seconds) but take a picture and compare the result to the other build methods. How would you count defects here? Is it even possible?
- Sometimes, the easiest way for materials scientists to start the building process is to gather all the atoms together, and then gradually rearrange them.
- Questions to consider. Look at your photos and Data Sheet. Compare the time and number of defects in each method.
- What did you notice about the number of defects in the various build conditions of Stage 2? Which method had the most? Which had the least?
- How did the number of defects compare to the time each one took to build?
- How did the number of defects compare to the level of control you had in placing atoms?
- Did certain methods tend to have particular types of defects?
- Did you discover any strategies for making fewer defects in any of the approaches?
These are questions materials scientists ask themselves after making a new material, too!
Clean-up:
Enjoy a piece of candy or two. Collect all candies and return them to their containers. Clean the work area and store the equipment properly.
Scoring Guide
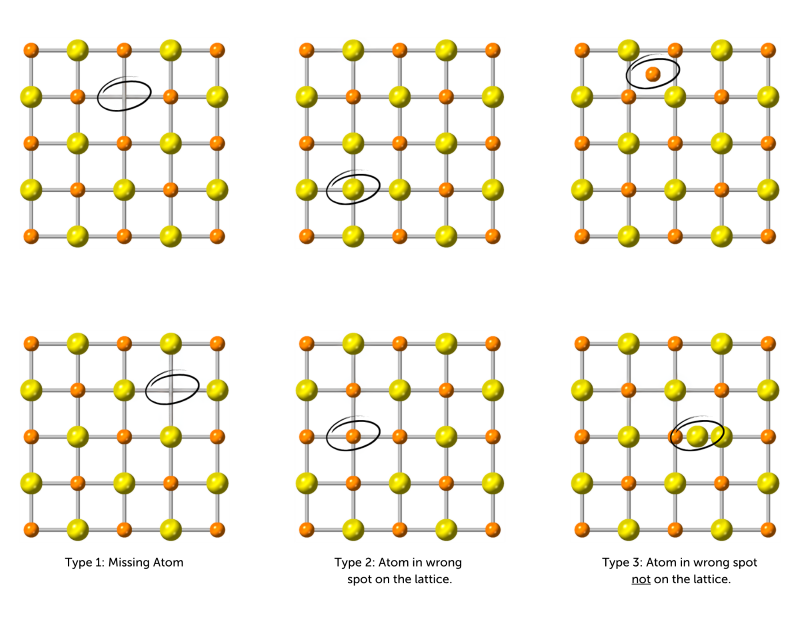
There are three types of situations where atoms find themselves misplaced from their intended location on the crystal lattice. These are called "defects."
Type 1: An atom is missing from its spot on the grid.
Type 2: An atom is in the wrong location on the alternating crystal lattice pattern.
Type 3: An atom is not in its proper location on the lattice.
After each build in Stage 2, look at your crystal structure to see how many times there is a match to one of these types. Count each example found as 1 defect.
-
What's Happening?
WHAT'S HAPPENING
How a material behaves is determined by how its atoms are arranged. So, materials scientists want to learn how different build methods affect a material’s resulting atomic structure. Looking for defects in an expected crystal structure gives them important clues.
You probably noticed different build methods led to different types and numbers of defects, especially when you tried to go fast. Materials scientists run into the same challenges when building crystals. Fast, high-energy processes, like making crystals with a laser, are rapid but result in more defects. Slow processes, like how crystalline rocks are formed deep within the Earth over time— usually under high pressure—have fewer defects.
Atoms are way too small to see or move around one-by-one like you did with your fingers, but new technologies give scientists better control of where atoms land. Then they use powerful microscopes to zoom in, see how the atoms are arranged, and analyze and interpret the image… just like you did!
-
So What?

Image credit: Adobe Stock
SO WHAT?Finding defects in predictable crystal patterns is a great way for scientists to learn about materials, but it’s also important for developing new technologies. For some applications, like materials used for computer memory, having very few defects is best. But defects aren’t always bad.
Scientists often want to add defects to a known structure to help produce a property they want. For example, steel is a material that needs to be extremely strong and rigid for many uses, but it also has to be bendable without cracking. Different methods and conditions for making steel change the arrangement of atoms inside, resulting in different properties of steel that can be used for everything from construction beams to washing machines.
-
Scientists In Action
Scientist in Action
Using plasma, scientists grow thin film crystals and use really cool equipment to study how tiny defects are made. In the process, they unlock new properties and even design entirely new materials. Meet Ben DeBastiani, Nathan McIlwaine, and Nestor Marquez Rios as they explore how to make these discoveries possible.
-
For Teachers
For Teachers
Below are suggested alignments with the Next Generation Science Standards. For the full list, please scroll down and download the PDF.
Performance Expectations
- 5-PS1-4 Conduct an investigation to determine whether the mixing of two or more substances results in new substances.
- 3-5-ETS1-2 Generate and compare multiple possible solutions to a problem based on how well each is likely to meet the criteria and constraints of the problem.
- 3-5-ETS1-3 Plan and carry out fair tests in which variables are controlled and failure points are considered to identify aspects of a model or prototype that can be improved.
- MS-ETS1-4 Analyze data from tests to determine similarities and differences among several design solutions to identify the best characteristics of each that can be combined into a new solution to better meet the criteria for success.
- MS-ETS1-4 Develop a model to generate data for iterative testing and modification of a proposed object, too, or process such that an optimal design can be achieved.
Disciplinary Core Ideas
PS1.B: Chemical Reactions
Grade 5
- When two or more different substances are mixed, a new substance with different properties may be formed.
ETS1.B: Developing Possible Solutions
Grade 3-5
- Research on a problem should be carried out before beginning to design a solution.
- Communicating with peers about proposed solutions is an important part of the design process, and shared ideas can lead to improved designs.
- Tests are often designed to identify failure points or difficulties, which suggest the elements of the design that need to be improved
Middle School
- A solution needs to be tested, and then modified on the basis of the test results, in order to improve it.
- There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem.
- Sometimes parts of different solutions can be combined to create a solution that is better than any of its predecessors.
- Models of all kinds are important for testing solutions.
ETS1.C: Optimizing the Design Solution
Grade 3-5
- Different solutions need to be tested in order to determine which of them best solves the problem, given the criteria and the constraints.
Middle School
- Although one design may not perform the best across all tests, identifying the characteristics of the design that performed the best in each test can proved useful information for the redesign process.
- The iterative process of testing the most promising solutions and modifying what is proposed on the basis of the test results leads to greater refinement and ultimately to an optimal solution.
Please click on the PDF below for a more detailed description of how this activity ties to NGSS
Download PDF


